

Inside our Manufacturing Environment
Standards & Certifications at Lúvacyt
%201.png)
%202.png)
What is an ISO Certified Cleanroom?
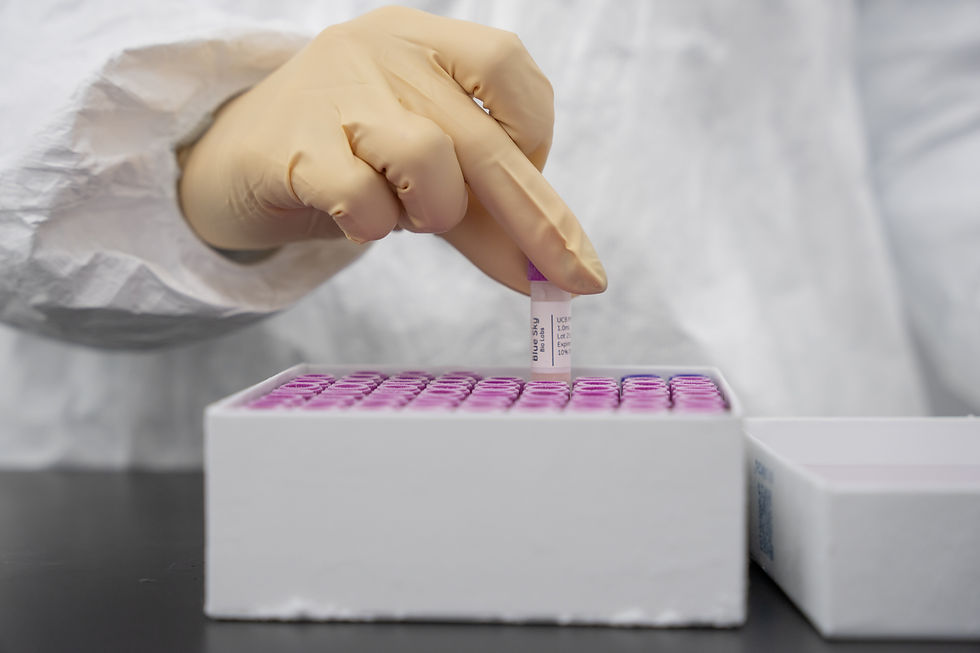
Part of the ISO cleanroom standards, ISO Class 5 is an extremely clean environment with strict limits on airborne particles. ISO Class 7 is a “clean but not aseptic-critical” environment. Often the surrounding room to an ISO 5 hood. Combined, these two standards provide some of the highest levels of cleanliness possible.
Why it matters:
Required for sterile operations like aseptic filling, cell isolation, exosome purification, and any step where open product exposure occurs.


%202.png)
%201.png)
Standards And Certifications
Luvacyt™ products are manufactured in a controlled laboratory environment designed to support consistency, quality, and regulatory compliance. Our facility utilizes ISO-classified cleanroom conditions, established quality management systems, and standardized processing protocols developed through decades of experience in cellular technologies.
Every stage of production is performed under defined environmental controls and documented procedures to support product integrity, traceability, and reliable manufacturing outcomes.


What is FDA Registered and GDUFA?
These approvals are an FDA regulatory framework requiring facilities involved in drug production or testing to register and pay user fees. It ensures transparency and regulatory oversight.
Why it matters:
Most small or mid-size biologics labs are not GDUFA registered. Being one sets us apart as a legitimate pharmaceutical-grade manufacturer, not a boutique stem cell shop.






What is an ISO Certified Cleanroom?
Good Laboratory Practices (GLP) is a regulatory framework (FDA, OECD) defining standards for non-clinical laboratory studies, including documentation, data integrity, training, equipment calibration, etc.
Good Manufacturing Practices (GMP) are FDA (and global) regulations governing how drugs, biologics, and medical products must be manufactured.
Why it matters:
GLP and GMP are the gold standards for manufacturing anything that could be used clinically – such as stem cells.




.png)